- Blog
- Sexy parodius pc download
- The last of us ed edd n eddy sound effects
- Stealth attraction words
- Minecraft optifine mod 1-8-9 forge
- Bluestacks download too many redirects
- Xerox Pws Release 8-1
- Sims 4 all expansions free download full version
- Eboot resigner 4-80
- Samsung se 208 driver windows 10
- Terraria maxed out character download
- Dog Honor Pathfinder Worksheet
- Nekojishi lin and partners download
- Skype download for windows 7 pc
- How to install dark souls 3 codex
- Business plan creator presentation software free download
- Disk defragmenter windows 10 stuck
- Tlc bone thugs concert tickets
- Intel centrino advanced n 6205 review
- The hammer driver
- Tangga lagu indonesia 2000
- Lego pirates of the caribbean codes xbox
- Endnote x7 mac free download
- Adobe premiere pro system requirements in virtualization
- The best calendar app mac pc ios app
- Touchcopy 12 torrent pirate
- Easy way to do dilutions
- Red hat linux ppp configuration

We add the ratio numbers: 20+1 and this gives us 21. So put 21.3oz of chemical in the container and fill the rest with water totaling 128oz to make a gallon of solution at a 5:1 dilution.
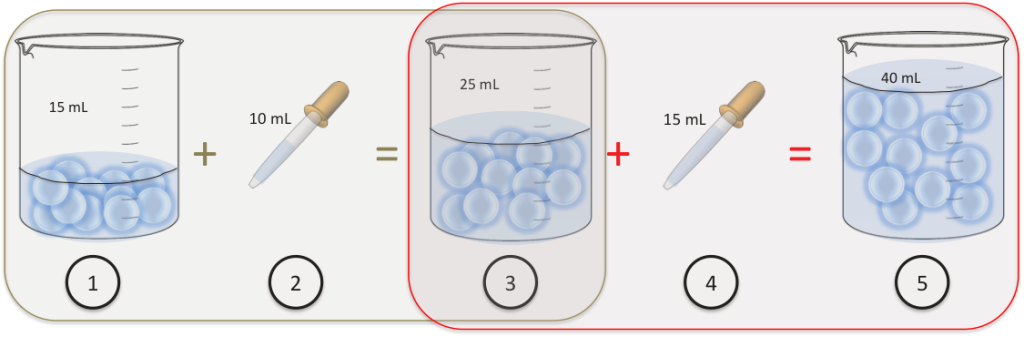
Then we take 128oz and divide that by 6 and we get 21.3333333. CALCULATE HOW MUCH SOLUTE YOU NEED (Translation: How much SOLID Kool-aid is needed ) Concentration g of solute/L of. To make a 5:1dilution ratio for a gallon, we add the ratio numbers together like this: 5+1=6. Problem: How would you prepare 100 mL of a 2.5 g/L solution of Kool-aid 1. However, the explanation of the dilution is often very short because there is limited space on. The process is exactly the same as the 32oz way. Many products are sold as 'concentrates', this means that you need to dilute them yourself before use. The first thing to know is that a gallon is 128oz. The dilution factor or the dilution is the initial volume divided by the final volume.
#EASY WAY TO DO DILUTIONS SERIAL#
So now lets say we want to use a gallon container. In serial dilutions, you multiply the dilution factors for each step.
#EASY WAY TO DO DILUTIONS HOW TO#
How to calculate Dilution Ratios of gallon containers. 10+1=11 Then 32oz divided by 11 = 2.9oz of chemical. How about a 10:1 dilution ratio for a 32oz bottle? It’s the same exact way. So put 4 ounces of chemical into the bottle and fill the rest with water for a 7:1 dilution. Again, change the dilution ratio numbers to addition like this: 7+1=8. Now lets do a 7:1 dilution for a 32oz bottle. 6.4 x 4 = 25.6, now we need to add back the one, which is the 6.4 and we get 25.6 + 6.4 = 32. So this means that we would need to put in 6.4oz of chemical and then fill the rest with water to make a 4:1 dilution ratio in a 32oz bottle. So for example: a dilution ratio of 4:1 would be 4+1=5 then I take the total ounces, which in this case is 32 and divide that by 5. The way that I do this is that I simply add the ratio numbers together. A serial dilution is the repeated dilution of a solution to amplify the dilution factor quickly.

We know that 32oz is the total ounces that we can put into that bottle, so lets figure out what parts need to be chemical and water. A dilution in chemistry is a process that reduces the concentration of a substance in a solution. Lets say we want to make a 32oz bottle with a 4:1 dilution (4 parts water and 1 part chemical). Over 1000 easy toddler activities that you can do at home. How to calculate dilution ratios of 32 oz bottles? To start with you need to decide what dilution ratio you are needing and what you are going to be putting it into, like a 32oz bottle, 128oz gallon jug, or any other containers that you know the ounces of.
Hopefully this helps, at least a little, for those who struggle with it or helps someone looking for an easier way than what they are using. With having to mix chemicals all the time, it was a pain. 12X 3) Isolate for "X": 120Ĥ) Solution: 1000 = X You would need to add 1000ml of sterile water.So this is something that I used to struggle with constantly. This way of expressing a dilution as a ratio of the parts of solute to the total number of parts is common in biology. Want to see the proper algebra way to do this problem in text? Question: The pharmacist hands you a 1.5L bottle of a 20% solution and asks that you mix it with sterile water to make as much 12% solution as possible. This type of pharmacy tech math may be more difficult for those who have not taken algebra in school or pharmacy dilution math before. If you want to cross check your answer, you could by taking this new information and doing an 2500mL - 1500mL - 1000mL - would be added to the solution.
